Clinical Trials
We started the VODCA project 10 years ago with the goal to create a software package specifically developed for the administration, organisation and analysis of multicentre clinical trials in radiotherapy. All the new features to better integrate the package into multi-vendor radiotherapy departments are also a great improvement for the use of VODCA in clinical trial.
The VODCA package supports centralised and decentralised, or federated, clinical trials and data collection.
Here are listed some useful features of VODCA when using the package for clinical trials:
- VODCA databases can be saved locally or remotely on PC and servers supporting all OS,
- VODCA databases are very easy to backup, copy, merge and share as a template,
- VODCA databases can be easily anonymised,
- Concurrent users can access VODCA databases,
- Data import has been validated through extensive use by a very large VODCA user community, and
- API (Application Programming Interface) for research and ad-hoc analysis is available.
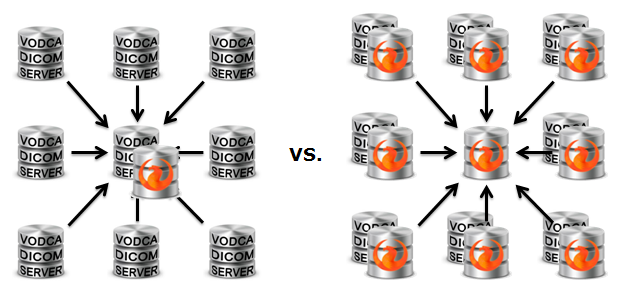
Centralised vs. decentralised clinical trial organisation
VODCA supports both approaches. However, we are convinced that decentralised clinical trials are the right solution in most of the cases! In fact:
- Decentralised clinical trials help building local knowledge in data collection, cleaning, analysis, archiving, organisation, publishing, distribution, curation, ...
- From a software perspective, developing a cloud strategy is all about the data and not moving it. VODCA has been developed following the basic principle of
"Taking the analysis to the data not the other way round!"
- There is also the topic of data security to consider - and that is a big topic! All this patient data movement, unloading and reloading provides numerous opportunities for security breaches.
- Multicentre clinical trials should centralise the local analysis results and not the data. Minimise the exchange of critical data!
- The harmonisation of the analysis methods in clinical trials is much more important than the database harmonisation!